|
12/9/2023 0 Comments Vsepr shapes and polarity 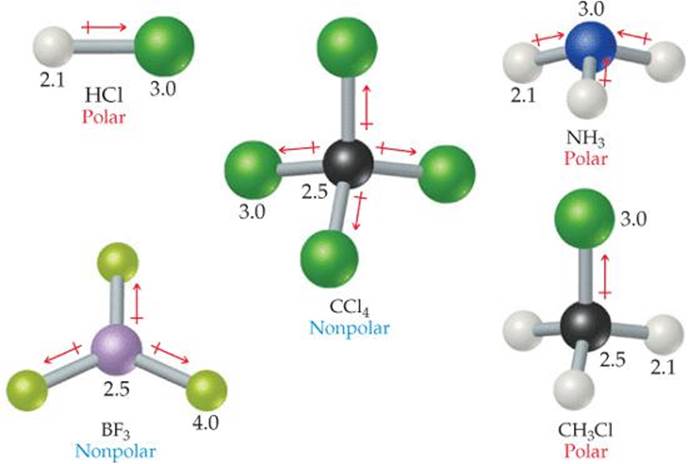 
You might already know that covalent bonds are typically represented as a line between two atoms. How do you draw the shapes of molecules in 3D?īefore we dive into examples of covalent structures, let's first learn about how we can represent them. By predicting the positions of electron pairs, we can predict the shape of simple covalent compounds, such as water and carbon dioxide. In summary, VSEPR is an important theory in chemistry that helps us to understand the shapes of molecules. If you want to refresh your memory on how atoms share electrons to achieve stable electron configurations, check out Covalent and Dative Bonding. By understanding the behaviour of electrons, we can predict the shape of simple covalent compounds. This helps to minimise repulsion and keep the molecule stable. So, VSEPR states that electron pairs repel each other and try to take up positions as far apart as possible. In simple covalent molecules, electron pair repulsion determines the position of the bonds, which in turn dictates the shape of the molecule. This is where the valence shell comes in - it's the outer shell of electrons in an atom. So, they try to stay as far away from each other as possible. Here's how it works: electrons like to move around in pairs, and because they are charged particles, they repel each other. This theory is used by chemists to predict the shape of molecules. To understand this, we need to talk about VSEPR, which stands for valence shell electron pair repulsion theory. Carbon dioxide has a straight line of atoms, while water is bent. Both of them are partially made of oxygen and contain covalent bonds. For example, water and carbon dioxide are two triatomic molecules that share some similarities. In the world of chemistry, we can find different shapes of molecules. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
